More than 10 Phase 3 trials combining ICIs with other agents have failed to deliver meaningful survival benefits.
These strategies all share the same limitation:
they act on adaptive immunity, while neglecting the upstream innate signals required to sustain anti-tumor responses.
The result:
This is not a checkpoint problem.
It is an innate immunity problem.
Effective immunotherapy requires early activation of innate immune pathways to:
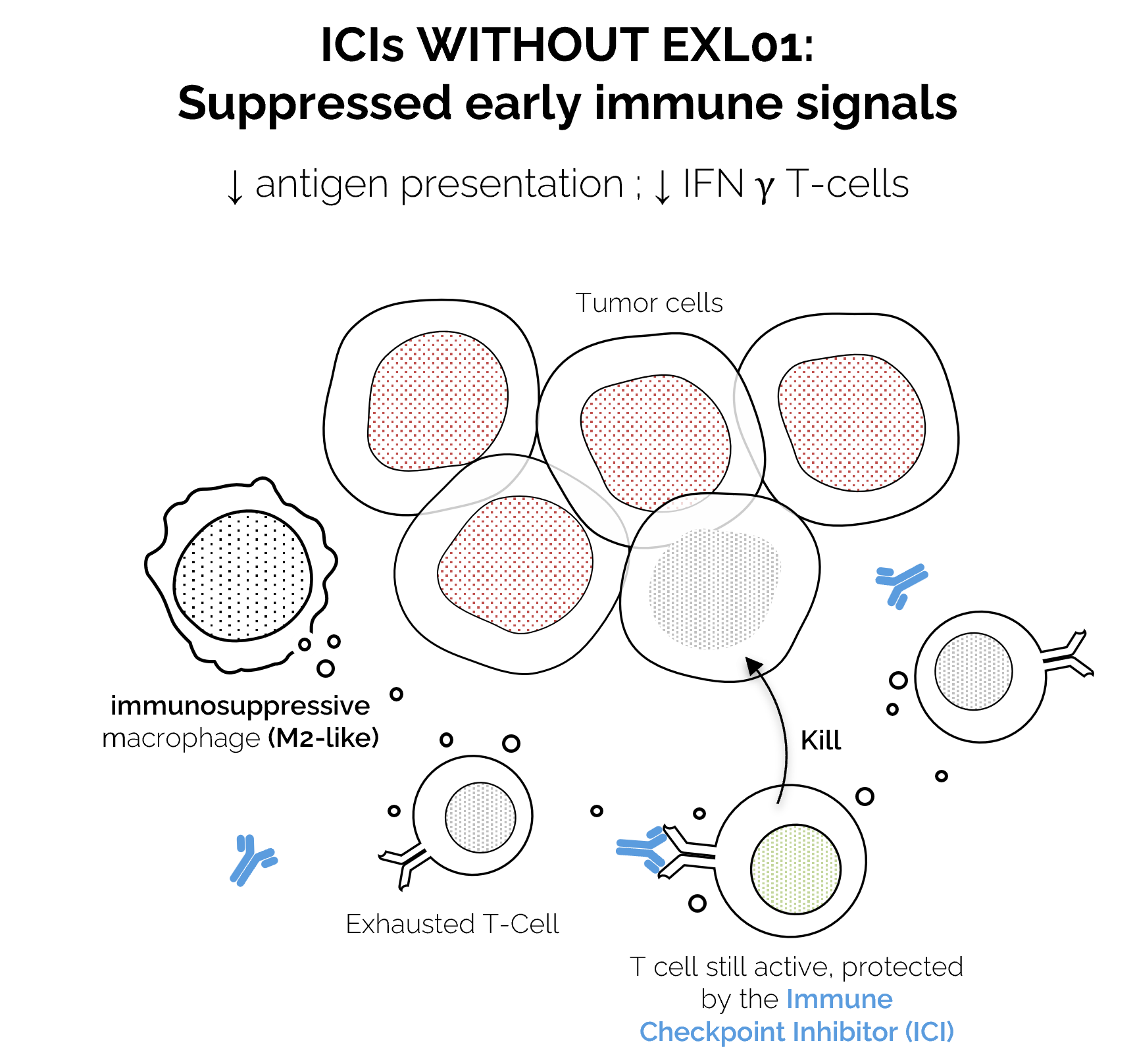
In many tumors, this layer is suppressed.
Restoring it is the key to breaking the ICI ceiling.
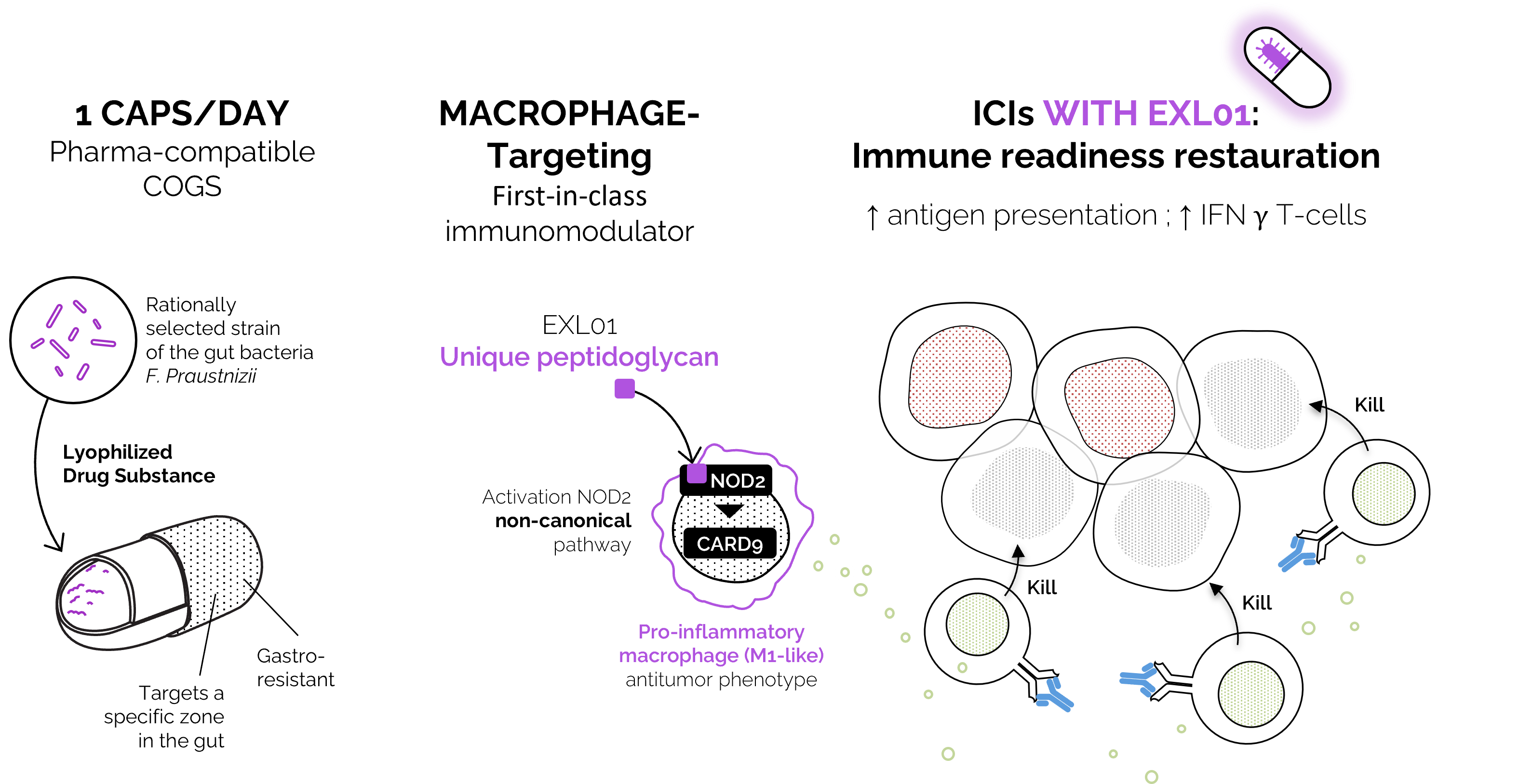
EXL01 is a macrophage-targeting immunomodulator that restores upstream innate immune signaling through activation of the NOD2–CARD9 pathway.
By reprogramming immunosuppressive macrophages, in the presence of checkpoint inhibitors, into pro-inflammatory, tumor-fighting cells, EXL01:
EXL01 acts upstream of the checkpoint - and is ICI-agnostic by design.
EXL01 is the first clinical-stage program targeting innate immune conditioning via the NOD2-CARD9 axis - the missing biological layer behind ICI resistance.
As ICI revenues approach $200B+, restoring response and durability represents a major strategic priority for oncology leaders.
EXL01 is positioned as:
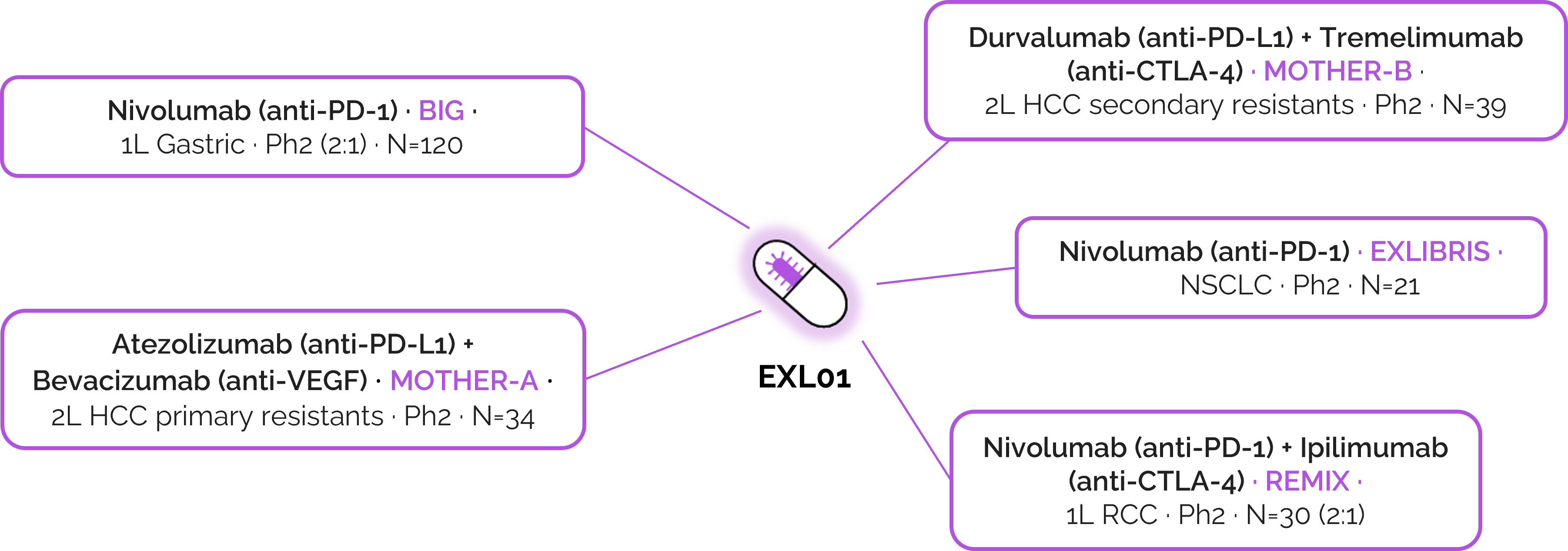
EXL01 is currently evaluated across multiple Phase 2 trials with leading ICIs:
A single mechanism, across multiple checkpoints and tumor types.
A key limitation in microbiome therapeutics has been industrialization — particularly for strict anaerobes.
Exeliom has solved this.
EXL01 is manufactured through a proprietary, vaccine-like GMP process enabling:
More than 10 GMP batches have already been produced, with successful process transfer across manufacturing sites.
This level of industrial maturity is rare in the live biotherapeutics field and removes a major barrier to late-stage development and commercialization.
EXL01 is not only biologically differentiated - it is industrially ready.
Beyond oncology, EXL01 leverages a unified biological mechanism to address:
This creates multiple value pathways from a single asset.
