EXL01 acts upstream of adaptive immunity through activation of the NOD2–CARD9
pathway, reprogramming macrophages and restoring the innate signals required for effective anti-tumor responses.
Because this mechanism operates independently of checkpoint blockade, EXL01 is inherently:
This positions EXL01 as a potential backbone therapy across immuno-oncology.
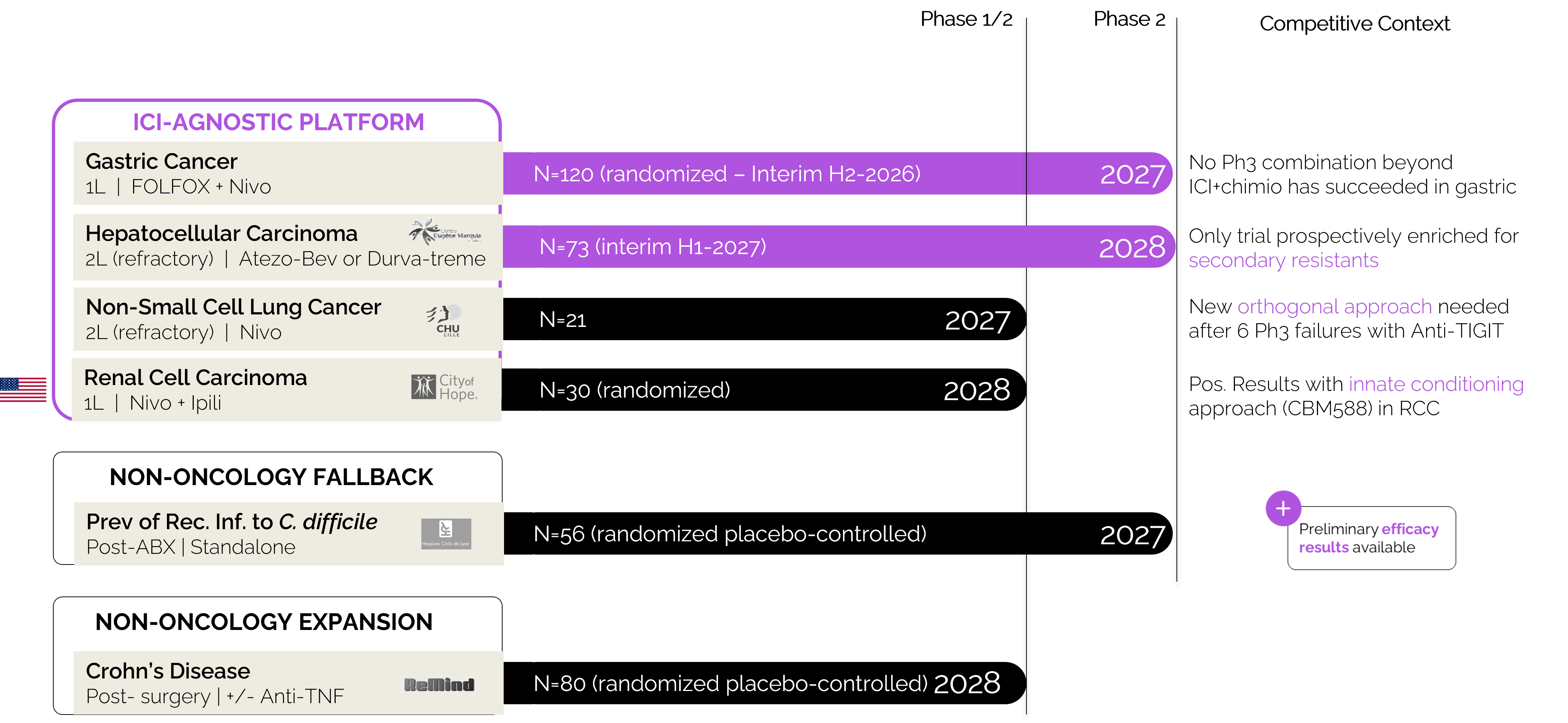
EXL01 is currently evaluated in combination with multiple checkpoint inhibitors:
These trials span:
EXL01 is the first clinical-stage program targeting innate immune conditioning across such a broad range of ICI combinations.
Ongoing clinical trials:
The BIG study is the lead value inflection point for EXL01.
This trial is designed to demonstrate that restoring innate immune activation can extend the durability of response to standard-of-care immunotherapy.
Interim readout: 2026
Topline results: 2027
BIG is not an isolated program - it is the first randomized validation of the platform.
EXL01 is also being evaluated in hepatocellular carcinoma in patients with primary and secondary resistance to immunotherapy.
NCT06551272
This program addresses a critical unmet need where current ICI-based strategies have limited efficacy.
By restoring upstream innate immune activation, EXL01 is designed to overcome resistance mechanisms that prevent durable responses.
Given the high unmet need and well-defined patient population, this study could support an accelerated development path, including adaptive Phase 2/3 strategies.
MOTHER represents a high-value, fast-to-signal opportunity within the EXL01 platform.
Beyond the lead program, EXL01 is being evaluated across multiple investigator-sponsored Phase 2 trials:
These studies:
This model enables rapid clinical breadth while maintaining a lean organization.
EXL01 also demonstrates activity outside oncology through the same biological foundation:
These programs provide additional value pathways while leveraging a shared platform.

